Read More
(1)(1)_w850_h1344_1.jpg)
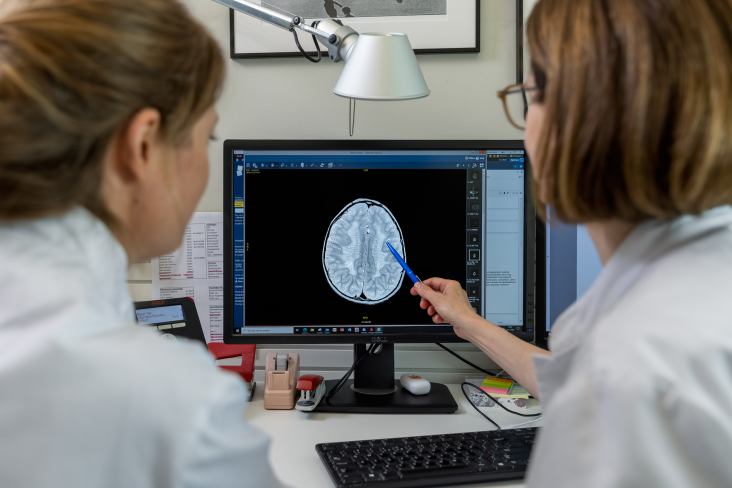
“With regards to the disease metachromatic leukodystrophy, or MLD, we see four to five new patients every year,” says Nicole Wolf, child neurologist at the Emma Kinderziekenhuis and professor Therapy of Leukodystrophies at Amsterdam UMC. “And in our field, that counts as a lot. Which means that, with such low patient numbers, the treatment of MLD and other genetic disorders of the brain are, by definition, personalized medicine.”
Occurrence
The various leukodystrophies occur in one in 700 to 800 births in the Netherlands. In many cases, the patients do not get to grow old. In a few cases the disease develops into a chronic one and in even rarer cases the leukodystrophies can improve on their own accord. For the majority, however, causes and treatments need to be found. “Though many patients are no longer eligible for such treatment,” notes Wolf. “Often the disease is already too advanced when the parents first come to the doctor.”
Experimental drugs
Because leukodystrophies are, by their very nature, genetic disorders, Wolf and her colleagues are also looking for the solution, to a large extent, in genetics. “For example, we are looking at gene therapy, in which the affected gene is ‘repaired’, for example in combination with stem cell transplantation, in which the repaired gene is put back into the body. But several drugs normally used for other disorders are also under investigation. One such instance is a drug that treats iron accumulation in blood diseases, which, a colleague found, might also provide relief in Pelizaeus-Merzbacher disease. That’s a syndrome in which part of the white matter is not built up properly.” To investigate the effect of this drug in patients, a study will start soon at the Amsterdam Leukodystrophy Center.
N-of-1
Wolf states that research on such extremely rare diseases is inherently difficult. “If there are only one or, at most, a few patients with a particular variant of a disease, then statistics lead to a so-called N-of-1 trial. How are you going to prove efficacy in these cases? Perhaps you’ll get to treat a teenager who can still walk and there’s a medicine that could prevent them from ending up in a wheelchair sometime in the future. But that can never be proven in the traditional way with a placebo-controlled double-blind design. And for very slowly progressive disorders we do not have the means yet to evaluate efficacy of a treatment in a single patient.”
Collaboration
The only solution, Wolf emphasizes, is collaboration. “That is collaboration between doctors at home and abroad, to find enough patients and therefore enough information to run a clinical trial; but also – and above all – collaboration with patients. Patients must also provide their input for the outcome measures of a study. What is important to them? What should remain or become possible, thanks to a drug?”
Expensive treatments
The discussion about the price of medicines is also a difficult one when it comes to these rare diseases. “The system of Quality-Adjusted Life Years, the so-called QALYs that are used in other research, are hardly tenable,” says Wolf. “If you can successfully treat a baby, you have gained so many healthy years of life that pharmaceutical companies can, and will, charge sky-high prices for it. But as a society we need to have this discussion, because the first gene therapies and medicines are now on their way. In France, Germany and Italy, for example, MLD patients are already being treated with gene therapy, and this will hopefully soon be the case in the Netherlands as well.”
Progress
Wolf particularly wants to stress that in recent years, important steps have been made in research on leukodystrophies. “Whereas ten years ago, no genetic diagnosis could be made for half of the disorders, this is now the case for only 10% of the patients. And that genetic diagnosis is the important first step towards a solution.”
Photography: Marieke de Lorijn